* This product is for research use only. Not intended for use in the treatment or diagnosis of disease.
Transfection of primary cells has always been a big problem in laboratory routines. Conventional calcium hydrogen phosphate precipitation, liposome-based transfection, and DEAE-glucan transfection are not suitable for primary cells because of their very low efficiency and high toxicity.
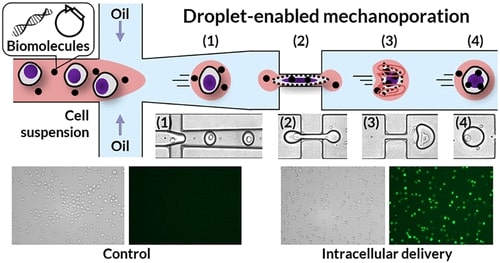
As an emerging gene delivery solution, microfluidics has been widely studied. Researchers have developed a new droplet squeezing platform that enables efficient, dose-controlled, and low analyte consumption delivery without the use of carriers or nanocarriers. The droplet microfluidics was combined with cell mechanoporation. Briefly, two immiscible fluids are used to first encapsulate single cells and exogenous molecules to produce oil-in-water droplets. These droplets then pass through a series of constrictions that deform the cell, creating discontinuities in the cell membrane (refer to this intracellular delivery method as "droplet squeeze"). In contrast to cell extrusion, due to the rapid cell deformation-recovery process, the recirculation flow formed in the droplet and the rigorous convective solution exchange achieved superior delivery performance, including plasmid DNA transfection. And can realize different large molecules (<2000 kDa glucan, 996 nt mRNA, and 7.9 kbp plasmid DNA) with high efficiency (up to 98%) and high throughput (up to 1 × 106 cells/min) of DNA delivery into various immune cells, including human primary T cells, with minimal cell disturbance.
A novel reagent is a biodegradable nucleic acid carrier consisting of a carbohydrate-based cationic polymer that combines with DNA to form nanoparticles. The complex enters the cell through endocytosis, where the cell takes up the biodegraded water unstable bonds, thereby releasing DNA in the perinuclear region of the cell for maximum gene expression. The advantages of low toxicity and efficient nuclear transport make it very suitable for the transfection of primary cells.
Electroporation is a physical method of gene delivery. Therefore, it is widely applicable to a variety of cell types, including animals, plants, and microorganisms. During electroporation, cells are exposed to high-voltage pulses in the presence of exogenous nucleic acids. The high voltage causes the cellular membrane to be transiently permeabilized, allowing exogenous nucleic acids (DNA or si RNA) to enter primary cells and other cells that are difficult to transfect. Several considerations need to be taken care of in order to optimize the delivery of nucleic acid into cells by electroporation. Electric field intensity and pulse duration are key parameters to maximize transfection efficiency and maintain cell viability. Buffer composition also affected transfection efficiency and cell viability. Traditionally, buffers with high ionic strength (low resistance), such as phosphate-buffered brine (PBS) or serum-free growth medium, have been used for electroporation of mammalian cells with high capacitance.
References: